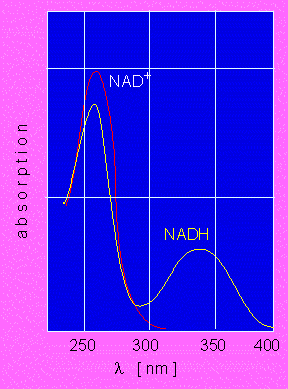
Optical Test According to O. Warburg. The reduced state of NAD (yellow curve) is characterized by an absorption of lambda = 340 nm. The oxydized state (red curve) displays no absorption at this wave length. In a given probe, the optical density (ordinate) is accordingly directly proportional to the amount of reduced NAD. The abscissa shows wave lengths between 250 and 400 nm.
The best-known and most common hydrogen acceptor is nicotinamide adenine dinucleotide (NAD):
NAD+ + 2e- + 2H+ <> NADH + H+
or, for example
NAD+ + R-CHOH-R' <> NADH + H+ + R-CO-R'
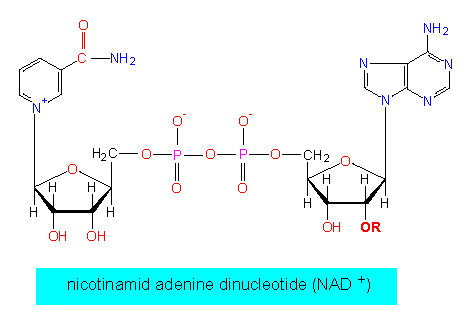
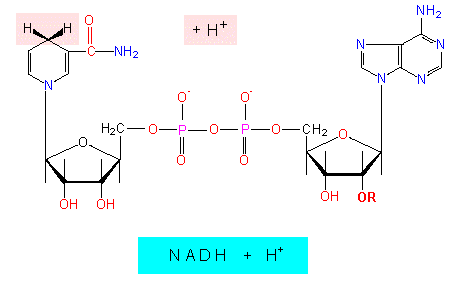
One of the protons is bound directly to the nicotine amide ring, while the other remains in solution.
NAD+ is a coenzyme (a prosthetic group). It has no effect on its own but functions only after binding to a protein. NAD+-binding proteins (enzymes) belong to the dehydrogenases. All of them catalyze the same chemical reaction (see above) though they differ with respect to their substrate specificity. Among many others, alcohol dehydrogenase, lactate dehydrogenase, malate dehydrogenase or glycerine aldehyde phosphate dehydrogenase are rather well known.
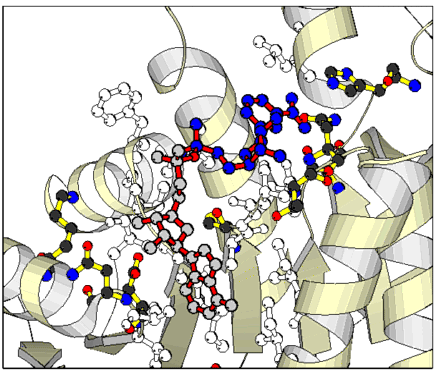
NAD - lactate dehydrogenase (LDH) - complex. The NAD (coloured) is bound in a bent conformation: Only part of the LDH enzyme is shown. The alpha-helices are displayed as bands, the beta - pleated sheets as arrows. Amino acid side chains that are in direct contact with NAD are outlined.
A derivative of NAD+ is NADP+ (nicotine amide dinucleotide phosphate). It is of special interest as it replaces NAD+ in photosynthesis. Both compounds are bound to their respective enzymes by weak interactions. They take on a specific conformation, when binding to a pocket at the enzyme's surface.
The delta G� of the reaction
NADH + H+ <> NAD+ + 2H+ + 2e-
is 14.8 kcal/mol (ca 62 kJ/mol), the E'� is -0.32. The signs change to the opposite, when the reaction runs the other way round.
Other hydrogen-transmitters are flavins, quinones, porphyrins (heme-groups) and several proteins containing metal-ions.
Flavins: The most common flavin is flavin adenine dinucleotide (FAD) that contains riboflavin or vitamin B2 besides an adenine residue.
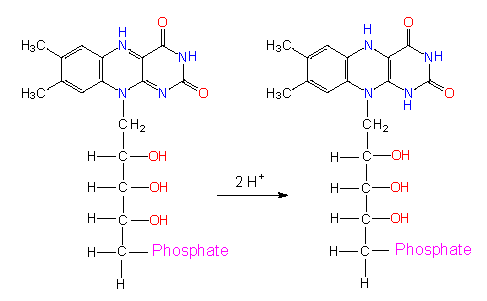
Riboflavin ( vitamin B2 ) in its oxydized and its reduced state
The term vitamin has actually nothing to do in a textbook on botany, because it stems from human nutrition physiology and comprises a group of molecules humans (and many animals) cannot synthesize. Accordingly, they do have to take it up with their vegetable nutrition. Most vitamins function as coenzymes. Since they are synthesized in plant cells, they should occur in the chapter on the molecular class they belong to. The active part of FAD is an alloxazin ring. In contrast to NAD+ (or NADP+), both protons and both electrons are bound by the ring system. In additin, FAD is bound covalently. This is also true for the related flavin mononucleotide (FMN). After binding of these cofactors, the enzymes take on a yellow colour. They are called flavoproteins. It should also be mentioned, that FAD and FMN are no nucleotides although their names imply it as the alloxazin ring is not bound to a sugar residue but to an alcohol (ribit, ribitol) instead. Among other reactions, flavins take part in the dehydrogenase reactions of the respiratory chain.
Quinones are involved in the redox reactions of mitochondria (respiratory chain) and chloroplasts (photosynthesis). It is distinguished between ubiquinones and plastoquinones. The two groups differ in their different side chains. Coenzyme Q, an unbiquinone, has a key position as a primary electron acceptor in photosystem II of photosynthesis.
Porphyrin rings and heme groups: Many
metals occur in more than one stable oxydized state. The
best-known and also most important example for cells is iron, that
occurs as trivalent FeIII and as bivalent
FeII.
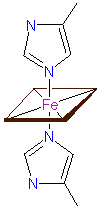
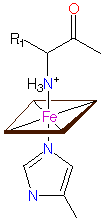
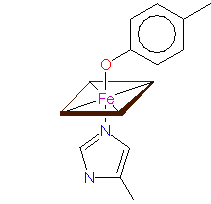
Heme-iron coordination - a.: in cytochrom c, cytochrom oxidase and others, b.: in cytochrom f, and c.: in cytochrom cd1 nitrite reductase © PROMISE
Only FeII, that has the ability to bind oxygen, occurs in the heme group (an iron-containing porphyrin ring) of hemoglobin (and leghemoglobin). In cytochromes, changes of valency occur. Cytochromes are members of both the respiratory chain and photosynthesis. The molecules differ mainly in their protein parts. The best-known metalloprotein without a heme group taking part in photosynthesis is ferredoxin.