As part of our discussion of organometallic reagents we compared the base strengths of methanide ion, amide ion, hydroxide ion, and fluoride ion. Figure 1 reiterates that comparison.
We also considered how it was possible to estimate the equilibrium constant for an acid-base reaction by comaparing the pKa values of the two acids involved in the reaction. Equation 1 presents another example. What is the approximate equilibrium constant for this reaction?
Within limits, it is possible to extend the use of pKa values to other reactions. Consider the addition of methyl lithium to formaldehyde as shown in Equation 2.
In this reaction, the negative charge starts out on a carbon atom and ends up on an oxygen. In terms of electronegativity, this is a favorable change. The alkoxide ion is a more stable species than the methanide ion. In other words, the alkoxide ion is a weaker base than the methanide ion. This means the equilibrium constant for reaction 2 will be greater than 1, i.e. at equilibrium there will be more product than reactant. How much more? To make a numerical estimate, compare the pKa values of the conjugate acids of the methanide and ethoxide ions in reaction 2; they are 50 and 16, respectively. Since methane is 1034 times less acidic than ethanol, methanide ion is 1034 times more basic than ethoxide ion. Hence the equilibrium constant for equation 2 is approximately 1034.
Now consider the reactions shown in Equations 3,4, and 5.
Estimate the equilibrium constant for each of these reactions. Is the addition of amide ion to a carbonyl group favorable? What about the addition of hydroxide ion and fluoride (and by extension, any halide ion)?
As this discussion indicates, the addition of anionic nucleophiles to the carbonyl group of aldehydes and ketones becomes less favorable as the nucleophilic atom changes from C to N to O to F. In fact, when the nucleophilic atom is F, the equilibrium constant is so small that it is safe to say that fluoride ion, and by extension, any halide ion, does not add to the carbonyl group of aldehydes or ketones.
Up to this point we have considered the relative nucleophilicities of anionic nucleophiles. The emphasis on the word anionic is necessitated by the fact, unlike ammonia, water, and hydrogen fluoride, methane does not have a lone pair of electrons on the central atom. In other words, methane is not nucleophilic. The methanide ion is. But ammonia and water, and by extension, amines and alcohols, can act as neutral nucleophiles. And under the right conditions they do.
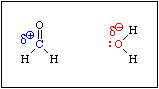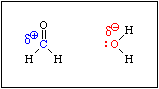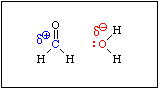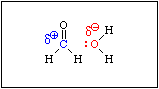
When formaldehde is added to water, the equilibrium shown in Equation 6 is established rapidly. An aqueous solution of formaldehyde is called formalin. It contains virtually no free formaldehyde, i.e. Keq >>> 1.
This reaction is analogous to the hydration of an alkene. If you recall, the mechanism of the acid catalysed hydration of an alkene involves protonation of the pi bond to produce an intermediate carbocation. Acid is required because the C-C bond is not polar. There is no C-C bond dipole to exert a Coulombic attraction toward a water molecule. Protonation produces a positive charge on one of the carbon atoms, which then attracts a lone pair of electrons on the oxygen atom of a water molecule. Since the pi bond in formaldehyde is polarized, water will be attracted to the positive end of the C-O bond dipole without the need for an acid.
The same is true of other aldehydes and ketones. But, as the bulk of the substituents attached to the carbonyl carbon increases, access to that carbon becomes more difficult and the rate of addition becomes slower.
In the same way that acid catalyses the addition of water to an alkene, it will also increase the rate of addition of water (and alcohols) to an aldehyde or ketone. Figure 2 compares the two processes.
The reactions of aldehydes and ketones with alcohols parallel their reactions with water. Figure 3 illustrates both processes.
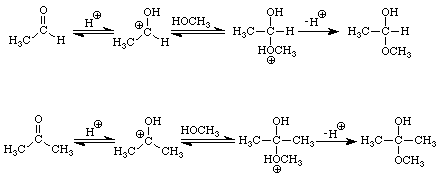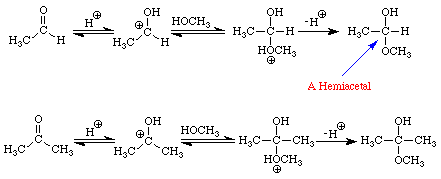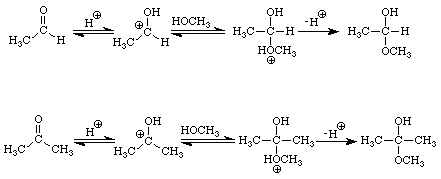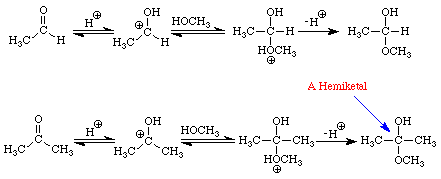
The addition of one molecule of an alcohol to an aldehyde produces a hemiacetal. Note that in acetaldehyde the carbonyl carbon has two bonds to the oxygen, one to the hydrogen, and one bond to carbon. In the hemiacetal the carbon also has two bonds to oxygen (different oxygens), one bond to hydrogen and one bond to carbon. The addition of one molecule of an alcohol to a ketone produces a hemiketal. The prefix hemi means half: under appropriate conditions, aldehydes and ketones may react with two molecules of an alcohol to produce acetals and ketals, respectively. Figure 4 extends the conversion of the methanol-acetaldehyde hemiacetal to its corresponding dimethylacetal. An analogous sequence of steps would convert the methanol hemiketal of acetone into the dimethylketal.
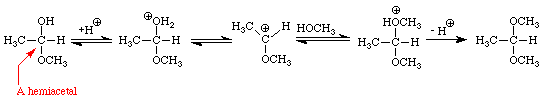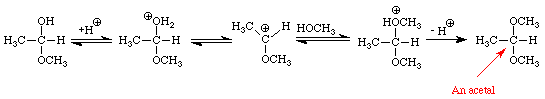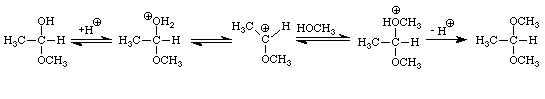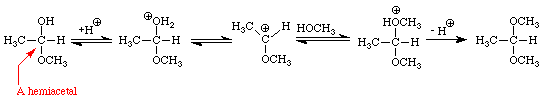
Many important examples of hemiacetals, acetals, hemiketals, and ketals may be found in sugars. The four sugars shown in Figure 5 contain examples of hemiacetals, acetals, and ketals.
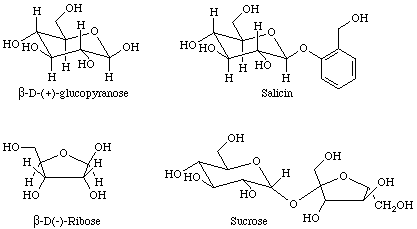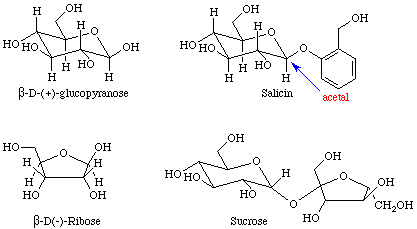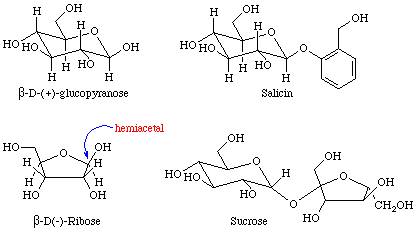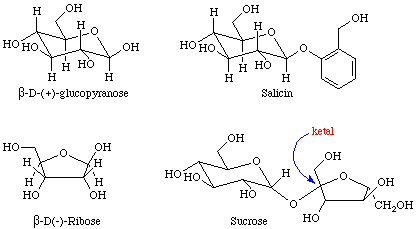
Equation 7 depicts the esterification of an alcohol that was performed during the total synthesis of taxol. Other, potentially reactive, OH groups were protected from reaction by the formation of a cyclic ketal prior to the esterification step.
The molecular fragment indicated in red in Equation 7 is part of a cyclic ketal, which, as Equation 8 indicates, may be formed by the reaction of a diol with a ketone.
The equilibrium shown in this reaction may be shifted to the right by use of an excess of alcohol or by removal of the water as it forms. Conversely, it may be shifted to the left, i.e. the diol may be regenerated from the cyclic ketal by addition of excess aqueous acid.